|
Cytoplasmatic proteins
that are involved in signal transduction are commonly constructed of modular
domains that either have a catalytic function (e.g. kinases) or belong to the
protein interaction domains (PIDs). The latter play a critical role in the
assembly of protein complexes that control the complex cellular behavior. One
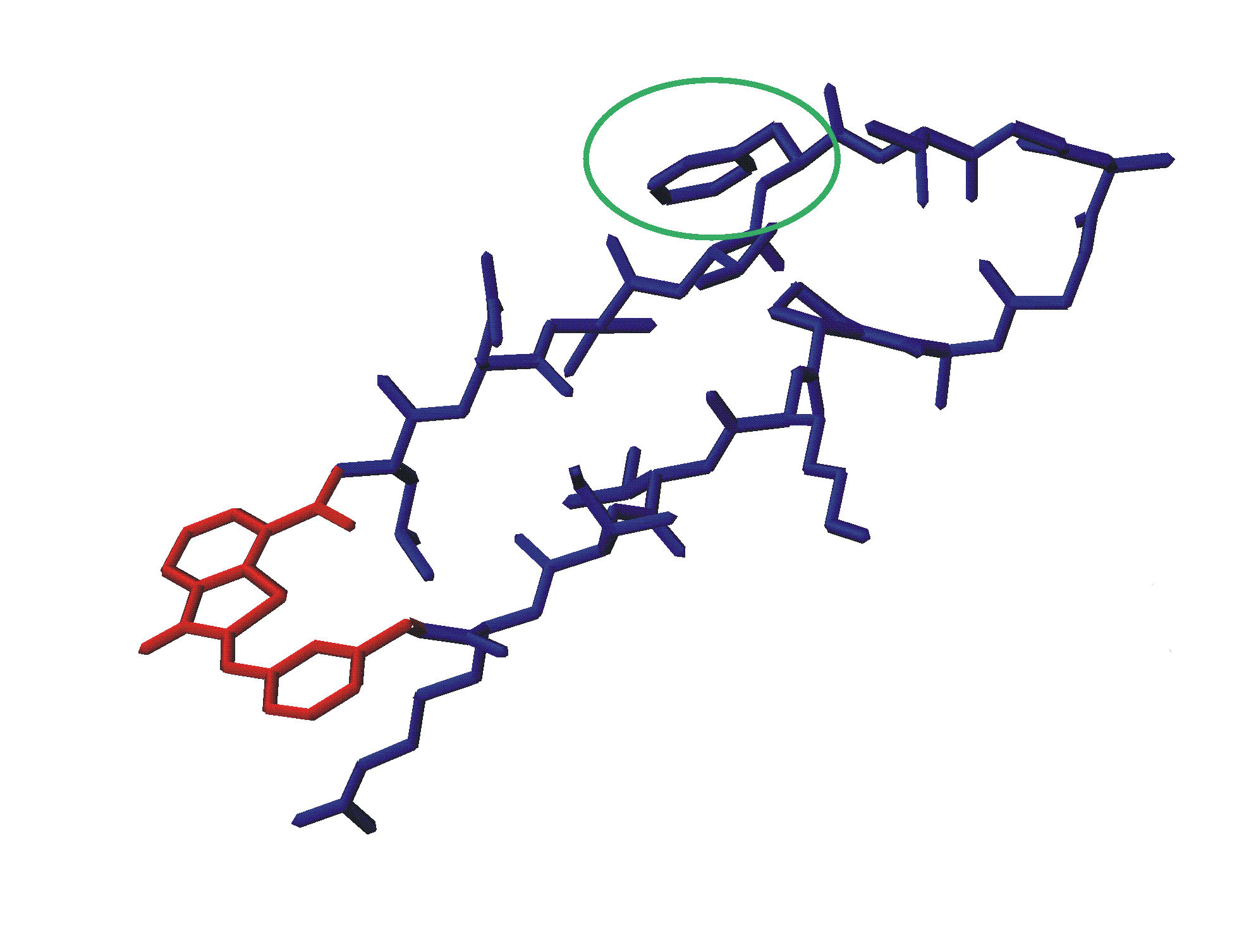
PID of great interest is the PDZ domain, a 100 amino-acid-residue domain. It
frequently occurs as multiple copies in cytoplasmatic proteins and is often
involved in the assembly of molecular complexes near the plasma membrane. While
most PDZ-domains recognize short, C-terminal peptide motifs, some do also
recognize internal binding motives like loops. The present project aims at the
design of ligands for both types of PDZ binding modes, using the syntrophin PDZ
domain as the protein partner. The ligands will be cyclic peptides that contain
a photosensitive, artificial amino acid. This will allow a control of
conformation and thus activity of the peptide ligands via the irradiation of
light. The artificial amino acids as well as the peptides will be synthesized,
the PDZ domain will be expressed in E.coli, the binding of the ligands to the
PDZ domain will be quantified and the structures of the free ligands and the
ligand-protein complexes will be determined using NMR
spectroscopy. |