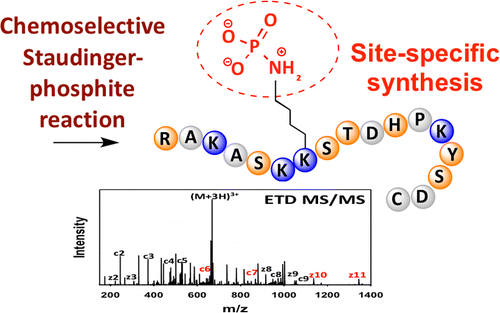
Site-specifically phosphorylated lysine peptides
J. Bertran-Vicente; R. Serwa; M. Schuemann; P. Schmieder; E. Krause; C.P.R. Hackenberger*
J. Am. Chem. Soc. 136, 13622-13628 (2014)
Protein phosphorylation controls major processes in cells. Although phosphorylation of serine, threonine, and tyrosine and also recently histidine and arginine are well-established, the extent and biological significance of lysine phosphorylation has remained elusive. Research in this area has been particularly limited by the inaccessibility of peptides and proteins that are phosphorylated at specific lysine residues, which are incompatible with solid-phase peptide synthesis (SPPS) due to the intrinsic acid lability of the P(?O)-N phosphoramidate bond. To address this issue, we have developed a new synthetic route for the synthesis of site-specifically phospholysine (pLys)-containing peptides by employing the chemoselectivity of the Staudinger-phosphite reaction. Our synthetic approach relies on the SPPS of unprotected e-azido lysine-containing peptides and their subsequent reaction to phosphoramidates with phosphite esters before they are converted into the natural modification via UV irradiation or basic deprotection. With these peptides in hand, we demonstrate that electron-transfer dissociation tandem mass spectrometry can be used for unambiguous assignment of phosphorylated-lysine residues within histone peptides and that these peptides can be detected in cell lysates using a bottom-up proteomic approach. This new tagging method is expected to be an essential tool for evaluating the biological relevance of lysine phosphorylation.
