The increasing number
of antibiotic-resistant bacteria poses a serious threat to public health. New
bioactive compounds are therefore highly desirable as new leads for drug
development. Cyanobacteria have proven to be a rich resource of new structures
of various classes, among them cyclic depsipeptides. After the identification
of a biological activity and the isolation of a new compound, its structure has
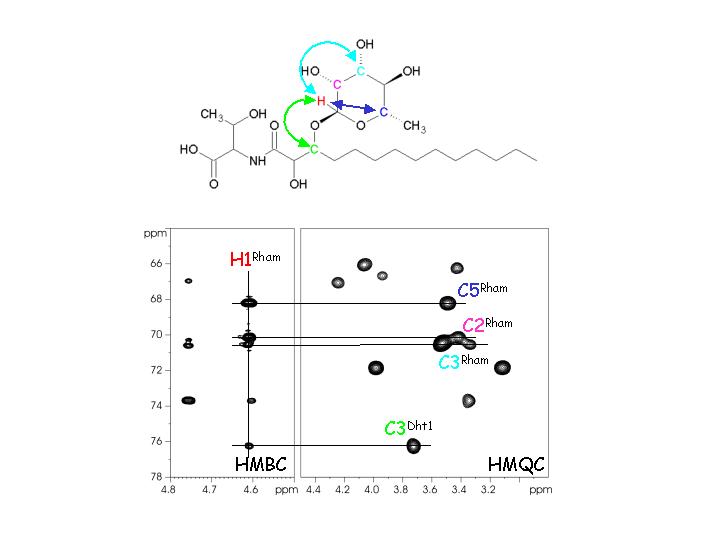
to be elucidated. Multidimensional NMR is a powerful tool for the structural
elucidation of natural products in combination with other techniques like mass
spectroscopy and chromatography.
We use modern, multidimensional NMR
techniques to elucidate the structure of novel compounds from the
cyanobacterium Hassallia sp., that turned out to be cyclic depsipeptides
containing both a lipid chain and a carbohydrate moiety.